Difference between revisions of "Creatinine"
Techsensus (talk | contribs) (Created page with "== Structure, properties and matrices == thumb|250px|Tautomer forms of creatinine '''Creatinine''' is an organi...") |
(No difference)
|
Revision as of 12:57, 5 December 2017
Contents
Structure, properties and matrices
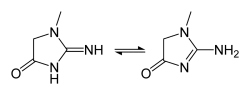
Creatinine is an organic compound characterized by the chemical formula C4H7N3O with a total molar mass of 113.12 g/mol and a density of 1.09 g/cm3. The solubility of creatinine in water is 90 mg/ml at 20°C. The molecule can tautomerize.[1] Anhydrous creatinine is a solid substance and generally considered as non-hazardous.[2]
Creatinine is the anhydride of its precursor molecule creatine (C4H9N3O2). Creatine is produced by liver, kidneys and the pancreas after which it is transported by the blood to other organs such as muscle and brain, where it is phosphorylated to the high-energy compound phosphocreatine involved in the metabolic process of muscle contraction. Creatinine is a breakdown product of phospocreatine and is normally excreted through the kidneys. Creatinine is usually produced at a fairly constant rate so that its equilibrium concentration is fairly constant in healthy individuals. The equilibrium concentration depends somewhat on diet and muscle mass, and therefore on age, sex, race and body build. Creatinine is commonly measured in blood and urine.[3]
Clinical significance
Creatinine blood levels often serves as a measure for determining the estimated Glomerular Filtration Rate (eGFR), which is a measure for renal function.[4][5][6] The GFR in mL/min can be calculated with the following formula: GFR = ( UX · V̇ ) / PX. Here, UX and PX are the concentrations of substance X in urine and plasma in mg/mL, respectively. V̇ is the urine flow in mL/min. Ideally X is a substance that is not altered, accumulated or resorbed by the kidneys and which has the same concentration in the plasma and glomular filtrate. Using creatinine in this formula, we obtain the creatinine clearance (CCr) resembling an eGFR. In the case of renal dysfunction, the creatinine clearance by the kidneys is reduced and therefore the creatinine concentration in the blood rises.[3]
Inulin is seen as the most reliable method for determining the GFR, measured by an intravenous injection and subsequent sampling of blood and urine. The calculated creatinine clearance turns out to be close to the clearance of inulin. In steady state the rate of urinary creatinine excretion is equal to the metabolic production of creatinine. Pathological conditions, such as hyperthermia, kidney failure or conditions of muscle wasting or damage, disturb the steady state. Overestimation of the GFR can occur at higher concentrations due to hyperexcretion. Consumption of meat, being rich in creatinine, also produces disturbances to the steady state condition.[3] Although the mentioned method for calculating the eGFR requires measurements of creatinine in urine, plasma concentrations of creatinine compared to normal ranges are also used for an indication of potential kidney failure.
Expected ranges of serum creatinine levels in healthy adults range from 0.6 to 1.2 mg/dL in males and 0.5 to 1.0 mg/dL in females.[7] [8] [9] Creatinine levels in infants and children are generally lower.[10] On the contrary, creatinine levels in elderly are often higher than average which leads to underestimation of their GFR when using creatinine serum levels as a measure.[11][12][13] Being a common biomarker for determining the GFR, the creatinine level is also often used for the dosing of some drugs.[11]
Surveys among general practicioners have indicated a significant desire for creatinine point-of-care tests. Out of 2770 respondents a total of 44% reported that they would use a creatinine point-of-care test.[14][15]
Lab protocols
Creatinine levels in blood are influenced by multiple anti-coagulants, of which most strongly by citrate. Heparin and EDTA show weaker effects.[16]
Creatinine is stable for at least 7 days in serum and urine at 4°C. In long term freezing at -20°C and below, serum creatinine is also stable, even after thawing and freezing. If serum and erythrocytes are not separated within 14 hours, the apparent creatinine concentration for some kinetic Jaffe assays is overestimated; this does not happen with enzymatic assays.[17]
It is important to control the temperature in order to minimize assay variations due to temperature.
State of the art sensing methods
Well established sensing methods of creatinine are chemical (Jaffe), enzymatic (creatininase), isotope-diluted mass spectrometry (IDMS) and high-performance liquid chromatography (HPLC).
The chemical Jaffe reaction is based on creatinine reacting with alkaline picrate, eventually forming an orange-red complex. This yields a measurable color change usable to determine the concentration of creatinine. A large drawback of this method is the low specificity for creatinine. The reaction has many interferrants such as glucose, bilirubin, pyruvate and guanidine. By tuning the reactant concentrations, reading time intervals, temperature and wavelength it is possible to increase the specificity of this method. The reaction is pseudo first order with respect to the picrate until 30 mmol/L. Methods generally use concentrations between 3 and 16 mmol/L, and above 6 mmol/L the color development becomes nonlinear. The reaction is also pseudo first order for hydroxide above 0.5 mmol/L. Above 200 mmol/L blank absorbance increases and above 500 mmol/L the Jaffe complex is degraded faster. For the Jaffe reaction the absorbance is maximal between wavelengths of 490 and 500 nm. Furthermore, temperature control is important since the temperature significantly influences complex formation and absorbance, in the range between 25°C and 37°C. So called compensation factors are often applied in order to account for interferences.[17]
Primarily, there are three approaches regarding the enzymatic methods. In the first method creatininase amidohydrolase is used to convert creatinine to creatine. The reaction is followed by those of creatine kinase, pyruvate kinase and lactate dehydrogenase. A decrease in absorbance at 340 nm is measured. The initial reaction step allows for prior removement of endogenous creatine and pyruvate. The required incubation time is about 30 minutes. The second approach starts with the conversion of creatinine to creatine using creatinase amidinohydrolase, sarcosine oxidase and peroxidase. The resulting hydrogen peroxide can be detected by a varity of methods. Care must be taken to prevent the inteference of bilirubin and ascorbic acid, e.g. by using bilirubin oxidase and ascorbate oxidase. Alternatively, one can use formaldehyde dehydrogenase instead of peroxidase and measure the reduction of NAD+. The last approach employs creatinine deaminase, L-methylhydantoinase, L-carbamoylsarcosine aminohydrolase, arcosine oxidase and peroxidase to, again, produce measurable hydrogen pyroxide.[17]
There are currently several commercially available biosensors that measure creatinine. Information is available of the i-STAT system made by Abbott, the StatSensor CREAT by Nova Biomedical[18], and the CardioChek PA by PTS Diagnostics. These sensors all make use of enzymes and electrochemistry to produce a current that linearly correlates with the concentration of creatinine.[19][20][21]
The numbers in the Table below have been collected per performance aspect. The commercial products combine certain aspects at lowest-performance and other aspects at highest-performance; none of the commercial devices achieve all aspects at highest performance or all aspects at lowest performance.
| Performance aspect | Low performance | High performance |
|---|---|---|
| Precision | 0.3 mg/dL[21] | 0.1 mg/dL[19] |
| Range | 0.2 - 10 mg/dL[21] | 0.2 - 20 mg/dL[19] |
| Sample volume | 20 µL[21] | 1.2 µL[20] |
| Speed | 8 min[21] | 30 sec[20] |
| Cost per instrument | € 12500[22] | € 400[20] |
| Cost per cartridge | € 9[23] | € 5[24] |
Note: Creatinine has a molar weight of 113 g/mol. Therefore, a concentration of 1 mg/dL corresponds to a molar concentration of 88.42 μmol/L.
Sensing methods in development
Novel methods are being investigated in order to improve e.g. the assay performance and cost effectiveness. The efforts are mainly oriented toward enzyme-free detection, e.g. by molecularly imprinted polymers (MIPs) or antibodies.
MIPs involve a polymer-based recognition motif that selectively interacts with creatinine, followed by colorimeteric or potentiometric detection.[25][26][27][28][29][30] Picric acid capped silver nanoparticles has been used as selective probes for colorimetric creatinine sensing.[31] Calix[4]arene thiol functionalized gold nanoparticles have been used to detect creatinine by means of dynamic light scattering. [32] Antibodies have been used to make a competitive creatinine immunosensor.[33]
Potential biomarkers for the future
| Name | Type |
|---|---|
| Calcium (Ca2+) | Electrolyte |
| Magnesium (Mg2+) | Electrolyte |
| Potassium (K+) | Electrolyte |
| Sodium (Na+) | Electrolyte |
| Bilirubin | Small molecule |
| Creatinine | Small molecule |
| Glucose | Small molecule |
| Lactate | Small molecule |
| Triglycerides (TG) | Small molecule |
| Urea | Small molecule |
| Uric Acid | Small molecule |
| Amino-terminal Propeptide of type I Collagen (PINP) | Protein |
| Amino-terminal Propeptide of type III Collagen (PIIINP) | Protein |
| Beta Human Chorionic Ganodotropin (β-hCG) | Protein |
| Brain Natriuretic Peptide (BNP) | Protein |
| Cardiac Troponin | Protein |
| Carboxy-terminal Propeptide of type I Collagen (PICP) | Protein |
| C-Reactive Protein (CRP) | Protein |
| Cross-linked Carboxy-terminal Telopeptide of type I Collagen (ICTP) | Protein |
| D-Dimer | Protein |
| Glycated Haemoglobin (HbA1c) | Protein |
| Haemoglobin (Hb) | Protein |
| High-Density Lipoprotein (HDL) | Protein |
| Low-Density Lipoprotein (LDL) | Protein |
| Metalloproteinase (MMP) | Protein |
| Thyroid Stimulating Hormone (TSH) | Protein |
| TIMP Metalloprotease Inhibitor (TIMP) | Protein |
| Chlamydia | Pathogen |
| Gonorrhea | Pathogen |
| Group A Streptococcus (GAS) | Pathogen |
| Helicobacter Pylori | Pathogen |
| Influenza | Pathogen |
| Methicillin-Resistant Staphylococcus Aureus (MRSA) | Pathogen |
| Vancomycin-Resistant Entrococcus (VRE) | Pathogen |
| White cell count | Others |
| International Normalized Ratio (INR) | Others |
| Erythrocyte Sedimentation Rate (ESR) | Others |
References
- ↑ Creatinine (last modified 23 March 2016), Wikipedia (Encyclopedia), retrieved from https://en.wikipedia.org/wiki/Creatinine, consulted on 12 May 2016
- ↑ SAFETY DATA SHEET Creatinine, Sigma-Aldrich, 21 December 2015, retrieved on 29 may 2016 from http://www.sigmaaldrich.com/MSDS/MSDS/DisplayMSDSPage.do?country=NL&language=EN-generic&productNumber=C4255&brand=SIAL&PageToGoToURL=http%3A%2F%2Fwww.sigmaaldrich.com%2Fcatalog%2Fproduct%2Fsial%2Fc4255%3Flang%3Den
- ↑ 3.0 3.1 3.2 Medical Physiology: A Cellular and Molecular Approach, updated second edition Walter F.Boron, Emile L. Boulpaep, 2012, Elsevier saunders, pages 767-770
- ↑ Renal function (last modified 26 May 2016), Wikipedia (Encyclopedia), retrieved from https://en.wikipedia.org/wiki/Renal_function, consulted on 29 May 2016
- ↑ Prediction of Creatinine Clearance from Serum Creatinine, Cockcroft D.W. and Gault M.H., 1976, Nephron, Volume 16, No. 1
- ↑ Predicting Glomerular Function from Adjusted Serum Creatinine, Gault M.H., Longerich L.L., Harnett J.D., Wesolowski C., 1992, Nephron, Volume 62, No. 3
- ↑ Reference Range and Method Comparison Studies for Enzymatic and Jaffé Creatinine Assays in Plasma and Serum and Early Morning Urine, Mazzachi BC, Peake MJ, Ehrhardt V., Clin Lab 2000;46:53-55.
- ↑ Reference Intervals for Serum Creatinine Concentrations: Assessment of Available Data for Global Application, Ferruccio Ceriotti, James C. Boyd, Gerhard Klein, Joseph Henny, Josep Queralto, Veli Kairisto, and Mauro Panteghini,7 on behalf of the IFCC Committee on Reference Intervals and Decision Limits (C-RIDL), 2008,Clinical Chemistry 54:3, pages 559–566
- ↑ Serum creatinine levels in the US population: Third National Health and Nutrition Examination Survey, CA Jones, GM McQuillan, JW Kusek, MS Eberhardt, WH Herman, J Coresh, M Salive, CP Jones, LY Agodoa, December 1998, American Journal of Kidney Diseases, Volume 32, Issue 6, pages 992-999
- ↑ High sensitive CRP and creatinine: reference intervals from infancy to childhood. Schlebusch H, Liappis N, Kalina E, Klein G., J Lab Med 2002;26:341-346.
- ↑ 11.0 11.1 Renal handling of drugs in the healthy elderly Creatinine clearance underestimates renal function and pharmacokinetics remain virtually unchanged, D. Fliser, I. Bischoff, A. Hanses, S. Block, M. Joest, E. Ritz, E. Mutschler, May 1999, Europeon Journal of Clinical Pharmacology, Volume 55, Issue 3, pages 205-211
- ↑ Estimated glomerular filtration rate leads to higher drug dose recommendations in the elderly compared with creatinine clearance, J. Q. Hudson1, J. R. Bean1, C. F. Burger1, A. K. Stephens4 and M.S. McFarland, March 2015, International Journal of Clinical Practice, Volume 69, Issue 3, pages 313-320
- ↑ Serum Creatinine and Functional Limitation in Elderly Persons, Michelle C. Odden, Michael G. Shlipak, and Ira B. Tager, March 2009, Journals of Gerontology Series A: Biological Sciences and Medical Sciences, Volume 64A N0. 3,pages 370-376
- ↑ Current and future use of point-of-care tests in primary care: an international survey in Australia, Belgium, The Netherlands, the UK and the USA, : Howick J, Cals JWL, Jones C, et al., BMJ Open 2014, http://bmjopen.bmj.com/content/4/8/e005611
- ↑ Sneltesten in de huisartspraktijk Huidig gebruik en behoefte aan testen in de toekomst, Cals JWL et al., Ned Tijdschr Geneeskd. 2014;158:A8210
- ↑ Effects of common anticoagulants (heparin, citrate and EDTA) on routine plasma biochemistry of cattle, M. Mohri, H. Shakeri, S. Lotfollah Zadeh, 5 January 2007
- ↑ 17.0 17.1 17.2 Tietz Fundamentals of clinical chemistry and molecular diagnostics, Carl A. Burtis, David E. Burtis, 2015, Seventh Edition pages 365-368
- ↑ Evaluation of the Nova StatSensor® XpressTM Creatinine Point-Of-Care Handheld Analyzer, Cara Simone Kosack, Wim de Kieviet, Kubra Bayrak, Anastacija Milovic, and Anne Laure Page, April 2015, PLoS, Volume 10, No. 4
- ↑ 19.0 19.1 19.2 Creatinine/Crea information sheet, retrieved on 29 May 2016 from https://www.abbottpointofcare.com/support/technical-documentation/cartridge-test-information-sheets
- ↑ 20.0 20.1 20.2 20.3 StatSensor information sheet, Nova Biomedical, A. Menarini Diagnostics, retrieved on 29 May 2016 from http://shop.menarinidiagnostics.se/startsida/patientnars-system/view/productdetails/virtuemart_product_id/41/virtuemart_category_id/8.html, http://shop.menarinidiagnostics.se/files/downloads/STAT_SENSOR_Eng.pdf, http://www.novabio.us/statstrip-creatinine/
- ↑ 21.0 21.1 21.2 21.3 21.4 Creatinine test strips information sheet, PTS Diagnostics, Cardiochek PA, retrieved on 29 May 2016 from http://apps.ptsdiagnostics.com/pdf_files/creat/creatinine_english.pdf
- ↑ Fisher Scientific, https://www.fishersci.com/shop/products/abbott-i-stat-1-wireless-handheld-point-of-care-testing-system-2/p-4279669, consulted on 22 November 2015
- ↑ Central Infusion Alliance, Statsensor CREAT, Nova Biomedical, https://www.ciamedical.com/creatinine-test-strips-statsensor-50-cs.html, consulted on 29 May 2016
- ↑ CLIAwaived Inc., Cardiochek PA http://www.cliawaived.com/cf.inventory.htm?action=showinvone&invid=2352&head=PTS+Panels+Creatinine+Test+Strips&desc=The+PTS+Panels+Creatinine+Test+Strips+are+packaged+with+25+tests+per+box+and+include+a+color-coded+MEMo+Chip+that+contains+lot-specific+calibration+and+expiration+dating+information.+The+creatinine+test+strips+have+a+measurement+range+of+0.2+to+10+mg%2FdL+with+results+available+in+eight+minutes+from+a+small%2C+fingerstick%2C+whole+blood+sample+(20ul).++The+Creatinine+test+strips+are+for+professional+in+vitro+diagnostic+use+only+and+are+NOT+CLIA+WAIVED!++FDA-Cleared.+For+Professional+use+only+with+the+CardiChek+PA+Analyzer.++(REFRIGERATED+ITEM--PLEASE+SELECT+NEXT+DAY+AIR+SHIPPING+FOR+PRODUCT+STABILITY) , consulted on 29 May 2016
- ↑ Guinovart, T., Hernández‐Alonso, D., Adriaenssens, L., Blondeau, P., Martínez‐Belmonte, M., Rius, F. X., Ballester, P. (2016). Recognition and sensing of creatinine. Angewandte Chemie, 128(7), 2481-2486. doi:10.1002/ange.201510136
- ↑ Sharma, A. C., Jana, T., Kesavamoorthy, R., Shi, L., Virji, M. A., Finegold, D. N., & Asher, S. A. (2004). A general photonic crystal sensing motif: Creatinine in bodily fluids. Journal of the American Chemical Society, 126(9), 2971-2977. doi:10.1021/ja038187s
- ↑ Li, T., Chen, P., Nien, P., Lin, C., Vittal, R., Ling, T., & Ho, K. (2012). Preparation of a novel molecularly imprinted polymer by the sol-gel process for sensing creatinine. Analytica Chimica Acta, 711, 83-90. doi:10.1016/j.aca.2011.10.031
- ↑ Nanda, S., An, S., & Yi, D. (2015). Measurement of creatinine in human plasma using a functional porous polymer structure sensing motif. International Journal of Nanomedicine, 10, 93-99. doi:10.2147/IJN.S88378
- ↑ Wen, T., Zhu, W., Xue, C., Wu, J., Han, Q., Wang, X., Jiang, H. (2014). Novel electrochemical sensing platform based on magnetic field-induced self-assembly of Fe3O4@Polyaniline nanoparticles for clinical detection of creatinine. Biosensors & Bioelectronics, 56, 180-185. doi:10.1016/j.bios.2014.01.013
- ↑ C.S. Pundir, Sandeep Yadav, Ashok Kumar (2013), Creatinine sensors, Trends in Analytical chemistry, 50, 42-52
- ↑ Parmar, A. K., Valand, N. N., Solanki, K. B., & Menon, S. K. (2016). Picric acid capped silver nanoparticles as a probe for colorimetric sensing of creatinine in human blood and cerebrospinal fluid samples. Analyst, 141(4), 1488-1498. doi:10.1039/c5an02303c
- ↑ Sutariya, P. G., Pandya, A., Lodha, A., & Menon, S. K. (2016). A simple and rapid creatinine sensing via DLS selectivity, using calix[4]arene thiol functionalized gold nanoparticles. Talanta, 147, 590-597. doi:10.1016/j.talanta.2015.10.029
- ↑ Guha, S., Warsinke, A., Tientcheu, C. M., Schmalz, K., Meliani, C., & Wenger, C. (2015). Label free sensing of creatinine using a 6 GHz CMOS near-field dielectric immunosensor. Analyst, 14(9), 319-327. doi:10.1039/c4an02194k