Vancomycin
 [1] [1]
| |
| Drug Name | Vancomycin |
|---|---|
| Type | Small Molecule [2], Glycopeptide antibiotic [3] |
| Synonyms | Vancocin, Vancomicina, Vancomycin, Vancomycine, Vancomycinum [2] |
| Molecular formula | C66H75Cl2N9O24[1] |
| Molecular weight | 1449.265 g/mol [1] |
| Exact mass | 1447.43 g/mol [1] |
| Solubility | 0.225 g/L [1] in water |
| Half life | In normal renal patients: 4-6 hours [3] In anephric patients: 7.5 days [2][3] |
| Clearance | 0.058 L/kg/h mean renal clearance [2][3] |
| Decomposition | Emits toxic fumes of NO (nitric oxide) and Cl2 (chlorine), when decomposed with heat [1] |
Contents
History and Structure of Vancomycin
History
In the 1950s, only few options were available for the treatment of penicillin-resistant staphylococcal infections. To remedy this, Eli Lilly and Company started a program with the aim of discovering new antibiotics. Eventually vancomycin, first called compound 05865, was discovered in 1952 in a soil sample from the jungles in Borneo. The discovery was made by E.C. Kornfield, an organic chemist who worked at Eli Lilly [4][5].
Vancomycin, derived from the word “to vanquish", proved to be effective against most types of gram-positive bacteria [5]. The FDA approved the drug in 1958 [4], despite concerns about possible toxicity and the fact that impurities in the drug caused red man syndrome - a hypersensitivity reaction resulting in red flushing and erythematous rashes on the face, neck and torso of patients [6]. Eli Lilly marketed vancomycin hydrochloride under the name Vancocin, until the patent ran out in the early 80s at which point generic versions of the drug became available [4][5].
Structure
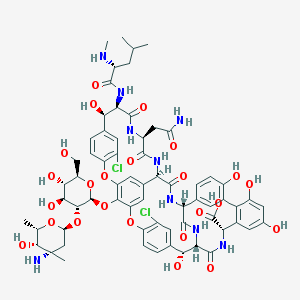
Vancomycin is naturally produced by the soil bacterium Amycolatopsis orientalis [7]. It is a glycosylated nonribosomal peptide, which means that it is biosynthesized by the bacterium without the use of mRNA or a ribosome, through means of nonribosomal peptide synthesis. It is a heptapeptide consisting of of the following amino acids: Leucine1 (Leu1), β-hydroxytyrosine2 (β-OH-Tyr2), Asparagine3 (Asn3), 4-hydroxyphenylglycine4 (HPG4), HPG5, β-OH-Tyr6, 3,5-dihydroxyphenylglycine7 (DPG7). Of these seven only Asn3 and Leu1 are proteinogenic amino acids, the rest being non-proteinogenic or non-coded amino acids. The stereoisomeric configuration of the amino acid residues in the heptapeptide scaffold of vancomycin is D-D-L-L-D-D-L [8].
Once the basic heptapeptide scaffold is assembled, further post translational modifications take place on the molecule. First, residues 2 and 4, 4 and 6, and 5 and 7, undergo oxidative crosslinking, become covalently bonded to each other, forming the highly rigid, dome-like structure of vancomycin. This conformation is what gives vancomycin its high affinity for forming hydrogen-bonds with its target - the N-acyl-D-Ala-D-Ala termini of the peptidoglycan precursors in bacteria. The molecule in this state is biologically active and is termed the aglycone backbone of vancomycin [9]. Finally, the Leucine is methylated to N-methylleucine, and two successive glycosylations on the phenolate of the HPG residue give the finished vancomycin molecule. These further modifications are not essential for the antibiotic functionality of vancomycin though they do allow for stronger interactions with the target [8].
Mechanism of action and antimicrobial resistance
Mechanism of action
Vancomycin kills and prevents the growth of gram-positive bacteria by inhibiting their cell wall synthesis [9]. The cell walls of gram-positive bacteria are comprised of several layers of peptidoglycan, a mesh-like polymer consisting of sugars and amino acids. This layer provides mechanical support so that these bacteria can withstand osmotic pressures as large as 5-15 atm without lysing (rupturing) [10].
A single peptidoglycan layer consists of many crosslinked glycan chains. A glycan chain is made up of repeating units of covalently bonded N-acetylglucosamine (NAG) and N-acetylmuramic acid (NAM) monomers joined together through transglycosylation. The newly elongated chains are mechanically weak until the pentapeptide chains on the NAM molecules are crosslinked. Crosslinking is effectuated by a family of transpeptidases, which use the amide group of the Lys3 on one strand to attack the D-Ala4 on the other strand, liberating a D-Ala5 residue, and forming a Lys3-D-Ala4 interstrand isopeptide bond which acts as a strengthening covalent cross-link between the two strands [10].
Vancomycin belongs to a class of antibiotics which interferes in both the polymerization and the cross linking of glycan strands. It does this by binding firmly to the substrate of the transpeptidation enzymes, the D-Ala4-D-Ala5 dipeptide, by means of five hydrogen bonds with its peptide backbone [9][11]. The formation of this complex prevents both transglycosylation and transpeptidation via steric hindrance [11].
The two final steps of bacterial peptidoglycan biosynthesis constitute a good target for any antimicrobial agent, as both processes are extracellular and thus accessible to compounds that are unable to penetrate the cell membrane. Furthermore, the peptidoglycan layer is so important for survival that it is highly conserved across organisms, meaning that compounds such as vancomycin are effective against a large variety of gram-positive bacteria. Lastly, targeting a process that involves multiple, related enzymes is advantageous as a single, spontaneous mutation in one enzyme will not lead to resistance [10].
Resistance to Vancomycin
Resistance to vancomycin and other GPAs (glycopeptide antibiotics) took over three decades to develop [10][7]. This exceptionally large delay between introduction into the clinic and the emergence of resistance, is due in part to the relatively low clinical use of vancomycin during the period following its introduction [7]. Indeed, once the first large outbreaks of β-lactam resistant strains of bacteria such as MRSA appeared in the 80s - causing a marked increase in vancomycin usage - vancomycin resistant bacterial strains quickly appeared as well [10][7].
Two distinct forms of vancomycin resistance exist. The milder form of vancomycin resistance, exhibited for example by VISA (vancomycin intermediate staphylococcus aureus) strains, develops in patients undergoing prolonged vancomycin therapy. The prolonged exposure to vancomycin puts selective pressure on the pathogens. The treatment turns a heterogenous colony of bacteria with only a small subpopulation having a vancomycin MIC (minimum inhibitory concentration) greater than 2 µg/mL, into a homogenous colony with a MIC of 8 µg/mL. The resulting colony becomes very difficult to eradicate with vancomycin therapy [12].
The second, more serious form of vancomycin resistance, demonstrated by bacterial strains such as VRSA (vancomycin resistant staphylococcus aureus), is not due to spontaneous mutations of pathogens upon continued exposure to the drug [10]. Instead, pathogenic microorganisms appear have directly copied the defense mechanisms of the antibiotic producing actinomycetes. This defense mechanism is used by the actinomycetes to avoid suicide during antibiotic production [10][7][13]. Pathogens resistant to GPAs obtain resistance through plasmid-borne copies of transposons coding for genes named van, which reprogram the biosynthesis of cell walls, replacing the D-Ala-D-Ala peptide terminus with a D-alanyl-D-lactate (D-Ala-D-Lac) terminus [13][10][14]. This small change reduces the binding affinity of vancomycin to the target around a 1000-fold, resulting in a vancomycin MIC ≥ 100 µg/mL making treatment with vancomycin impossible and effectively rendering the organism resistant [10][7][14].
Medical Use and TDM
Vancomycin is a glycopeptide antibiotic used as a last resort to treat severe, life-threatening infections caused by multidrug-resistant gram-positive bacteria, such as methicillin-resistant Staphylococcus aureus (MRSA) [13]. Vancomycin can be administered intravenously or orally. When taken orally, vancomycin is absorbed very poorly into the bloodstream [3]. Therefore, oral intake is only used for infections within the gastrointestinal tract, such as diarrhea caused by Clostridium difficile, and to treat enterocolitis caused by certain types of bacteria [15]. In all other cases vancomycin is administered intravenously [16]. Care must be taken to not administer the drug too fast, as this may lead to the patient developing red man syndrome [6]. Most hospital protocols recommended a minimum vancomycin infusion time of 60 minutes [6].
For patients with severe, deep-seated infections (including but not limited to meningitis, pneumonia osteomyelitis, endocarditis, bacteremia and prosthetic joint infection) a serum trough concentration of 15 to 20 µg/mL is recommended [17][18]. To achieve this concentration more rapidly an initial loading dose of 20-25 mg/kg is often given, followed by intermittent maintenance dosing of typically around 15-20 mg/kg every 8 to 12 hours [17]. Patients with less severe infections (soft tissue infections) do not require a loading dose, and are started immediately on intermittent dosing with the aim of achieving a minimum serum trough concentration of 10-15 µg/mL [18]. Trough concentrations are measured as these constitute the most practical and accurate indicator for treatment effectiveness and toxicity. Peak levels are rarely measured and are not clinically relevant [17].
| Target group | Loading Dose | Target Trough Concentration |
|---|---|---|
| Patients with deep-seated infections | 20 – 25 mg / kg | 15 – 20 µg/mL |
| Patients without severe infections | - | 10 – 15 µg/mL |
There is large interpatient variability in vancomycin pharmacokinetics [19]. The rate of clearance of vancomycin depends on the following factors; pathogen susceptibility to the drug, disease severity, the site of the infection, patient weight, age, gender and renal function [18]. As vancomycin is largely cleared through the kidneys, the effect of renal function is especially great. Groups of patients demonstrating rapid clearance, such as young children with naturally high renal function and burn patients, require significantly more frequent dosing than patients with normal renal function to achieve the same target trough concentrations [17][18][19]. In contrast, patients with impaired renal function may require dose reductions or extended dosing intervals in order to stay within the therapeutic range [17][18].
For optimal treatment it is essential that every patient receives the correct dose. If the concentration of vancomycin in the body is too low, the treatment is ineffective and the chance that bacteria develop resistance to vancomycin increases [12][20]. A concentration of vancomycin that is too high, has been linked to nephrotoxicity and ototoxicity [17].
Correct dosing of vancomycin is difficult due to the drug’s marked pharmacokinetic variability. Therapeutic drug monitoring (TDM) is the practice of measuring the concentration of a specific drug in the bloodstream at designated time intervals with the aim of using this data to optimize the individual dosing regimens of patients. TDM is of strong help for correctly dosing vancomycin [21]. Currently, hospital protocols recommend once-weekly monitoring of vancomycin levels for patients with stable renal function, and more frequent monitoring for those with changing renal function or those who are hemodynamically unstable [17]. The introduction of a biosensor for vancomycin would allow for more frequent monitoring and TDM for all patients which would contribute strongly to patient health and recovery.
Lab Protocols
Vancomycin hydrochloride may cause skin irritation, breathing difficulties and can be harmful if ingested. Protective eyewear, clothing and gloves have to be worn when working with the compound, and inhaling the dust/fumes/vapours/sprays of the compound is to be avoided [22].
Dry vancomycin hydrochloride powder should be stored at 20-25°C in order to be kept in optimal condition [3].
Vancomycin hydrochloride dissolved in human blood plasma should be stable for at least six months when stored at -80°C. Stable meaning that the solution loses less than 10% of its initial vancomycin concentration. With the incorporation of up to four freeze-thaw cycles it should be stable for at least four months if kept at -80°C. Vancomycin hydrochloride should furthermore be stable in plasma for at least 24 hours when kept at a temperature of 4°C [23].
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 PubChem (2005). Vancomycin compound summary for PubChem CID 14969. Accessed on 17 October 2017, at https://pubchem.ncbi.nlm.nih.gov/compound/vancomycin.
- ↑ 2.0 2.1 2.2 2.3 Drugbank (2005). Vancomycin. Accessed on 15 October 2017, at https://www.drugbank.ca/drugs/DB00512.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 Pfizer Labs (2010). Leaflet Sterile vancomycin hydrochloride, USP. Pfizer Inc.
- ↑ 4.0 4.1 4.2 Flynn Pharma LTD (2013). A short history of vancomycin.
- ↑ 5.0 5.1 5.2 Levine, D.P. (2006). Vancomycin: A History. Clinical Infectious Diseases, 42(1), 5-12. doi:10.1086/491709
- ↑ 6.0 6.1 6.2 Sivagnanam, S. & Deleu, D. (2003). Red man syndrome. Critical Care, 7(2), 119-120. doi:10.1186/cc1871
- ↑ 7.0 7.1 7.2 7.3 7.4 7.5 Grace, Y., Koteva, K.P., Thaker, M.N., Wright, G. (2014). Glycopeptide antibiotic biosynthesis. The Journal of Antibiotics, 67, 31-41. doi:10.1038/ja.2013.117
- ↑ 8.0 8.1 Nolan, E.M. & Walsh, C.T. (2009). How Nature Morphs Peptide Scaffolds into Antibiotics. Chembiochem : A European Journal of Chemical Biology, 10(1), 34-53. doi:10.1002/cbic.200800438
- ↑ 9.0 9.1 9.2 Reynolds, P.E. (1989). Structure, Biochemistry and Mechanism of Action of Glycopeptide Antibiotics. European Journal of Clinical Microbiology and Infectious Diseases, 8(11), 943-950. doi:10.1007/BF01967563
- ↑ 10.0 10.1 10.2 10.3 10.4 10.5 10.6 10.7 10.8 Kahne, D., Leimkuhler, C., Lu, W., Walsh, C. (2005). Glycopeptide and Lipoglycopeptide Antibiotics. Chemical Reviews, 105(2), 425-448. doi:10.1021/cr030103a
- ↑ 11.0 11.1 Van Bambeke, F., Van Laethem, Y., Courvalin, P., Tulkens, P.M. (2004). Glycopeptide Antibiotics: from Conventional Molecules to New Derivatives. Drugs, 64(9), 913-936. doi:10.2165/00003495-200464090-00001
- ↑ 12.0 12.1 Gardete, S. & Tomasz, A. (2014). Mechanisms of vancomycin resistance in Staphylococcus aureus. The Journal of Clinical investigation, 124(7), 2836-2840. doi:10.1172/JCI68834
- ↑ 13.0 13.1 13.2 Binda, E., Marinelli, F., Marcone, G.L. (2014). Old and New Glycopeptide Antibiotics: Action and Resistance. Antibiotics, 3(4), 572-594. doi:10.3390/antibiotics3040572
- ↑ 14.0 14.1 Miller, W.R., Munita, J.M., Arias, C.A. (2014). Mechanisms of antibiotic resistance in enterococci. Expert Review of Anti-Infective Therapy, 12(10), 1221-1236. doi:10.1586/14787210.2014.956092
- ↑ PubMed Health (2017). Vancomycin (By mouth). Accessed on 17 October 2017, at https://www.ncbi.nlm.nih.gov/pubmedhealth/PMHT0012602/?report=details.
- ↑ PubMed Health (2017). Vancomycin (By injection). Accessed on 17 October 2017, at https://www.ncbi.nlm.nih.gov/pubmedhealth/PMHT0012603/?report=details.
- ↑ 17.0 17.1 17.2 17.3 17.4 17.5 17.6 17.7 Consgrove, S.E., Avdic, E., Dzintars, K. & Smith, J. (2015). Antibiotic Guide. Johns Hopkins Medicine, The Johns Hopkins Hospital Antimicrobial Stewardship Program.
- ↑ 18.0 18.1 18.2 18.3 18.4 Drew, R.H. & Sakoulas, G. (2017). Vancomycin: Parenteral dosing, monitoring, and adverse effects in adults. Accessed on 26 September 2017, at https://www.uptodate.com/contents/vancomycin-parenteral-dosing-monitoring-and-adverse-effects-in-adults.
- ↑ 19.0 19.1 Carter, B.L., Damer, M.K., Walroth, T.A., Buening, N.R., Foster, D.R. (2015). A systematic Review of Vancomycin Dosing and Monitoring in Burn Patients. Journal of Burn Care & Research, 36, 641-650. doi:10.1097/BCR.0000000000000191
- ↑ Abdul-Aziz, M.H., Lipman, J., Mouton, J.W., Hope, W.W., Roberts, J.A. (2015). Applying Pharmacokinetic/Pharmacodynamic Principles in Critically Ill Patients: Optimizing Efficacy and Reducing Resistance Development. Seminars in Respiratory and Critical Care Medicine, 36(1), 136-53. doi:10.1055/s-0034-1398490
- ↑ Kang, J.S. & Lee, M.H. (2009). Overview of Therapeutic Drug Monitoring. Korean Journal of Internal Medicine, 24(1), 1-10. doi:10.3904/kjim.2009.24.1.1
- ↑ Cayman Chemical (2014). Safety Data Sheet Vancomycin Hydrochloride.
- ↑ Zhang, M., Moore, G.A., Young, S.W. (2014). Determination of Vancomycin in Human Plasma, Bone and Fat by Liquid Chromatography/Tandem Mass Spectrometry. Journal of Analytical & Bioanalytical Techniques, 5(3), 196. doi:10.4172/2155-9872.1000196